Scientists have long known that aging is not simply the passage of time, but a gradual loss of physiological reserve. Yet recent studies suggest a far more dramatic reality: for most people, health remains relatively stable until a sudden turning point in the mid-70s, when the balance between damage and repair abruptly collapses. This “tipping point,” identified through dynamical modeling of frailty, intersects with decades of research on cardiac aging and lung function trajectories, and together they reveal a deeply interconnected system in which inflammation plays a central role.
A Lifelong Tug-of-War Between Damage and Repair
The human body is engaged in a constant balancing act. Each day brings new forms of biological wear, cellular injury, oxidative stress, microinflammation, and each day the body mounts repairs. A study using data from two major longitudinal cohorts of older adults showed that this equilibrium holds remarkably well through early and mid-adulthood. But both robustness (the ability to resist new damage) and resilience (the ability to recover) steadily weaken over time. Around age 75, these two forces reach a point of balance, and then imbalance, where damage begins to outpace repair. Once this shift occurs, deficits accumulate rapidly, creating a steep rise in frailty and a measurable increase in vulnerability.
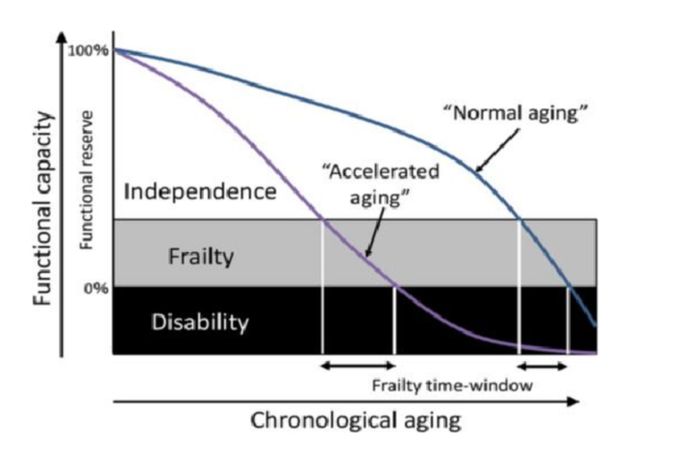
Frailty as the Clinical Expression of Declining Resilience
Frailty is not simply a marker of age, but a clinical syndrome characterized by diminishing strength, reduced endurance, and decreased physiological flexibility. It arises not from a single disease but from a tapestry of impairments across multiple systems. Clinically, frailty can be recognized through observable traits such as exhaustion, slowed walking, weakness, or unintentional weight loss. It can also be quantified through a Frailty Index, which measures the proportion of health deficits an individual has accumulated. As the tipping point approaches, the frailty burden accelerates, and once it crosses beyond a certain threshold, recovery becomes increasingly difficult. Small stressors, a mild respiratory infection, a brief hospitalization, may trigger disproportionate decline.
The Role of Cellular Senescence and Chronic Inflammation
Underlying the frailty tipping point is a biological phenomenon known as cellular senescence. As people age, more cells enter a state of permanent dysfunction in which they no longer divide but remain metabolically active. These senescent cells release inflammatory molecules collectively known as the Senescence-Associated Secretory Phenotype, or SASP. This chronic inflammatory milieu subtly alters the body's internal landscape: muscles lose mass more quickly, tissues repair more slowly, and the immune system becomes less coordinated. Over years, inflammation gradually erodes the body’s reserves. By the mid-70s, the cumulative effect becomes enough to push the system into imbalance.
The Aging Heart as an Early Indicator of Systemic Strain
The heart provides one of the clearest examples of this slow-building vulnerability. Even in healthy adults, the cardiac muscle becomes stiffer with age. The left ventricle thickens, diastolic filling slows, and the responsiveness to stress diminishes. At the cellular level, impaired autophagy allows damaged mitochondria to accumulate. Reactive oxygen species rise, and the heart’s ability to adapt becomes limited. These changes rarely cause symptoms early on, but they reduce the heart’s capacity to compensate for stressors. Consequently, heart failure and atrial fibrillation become far more common after age 75, often appearing abruptly rather than gradually.
The Lifelong Lung Trajectory That Shapes Aging
While frailty and heart failure tend to emerge later in life, the lungs follow a trajectory that begins much earlier. Lung function increases steadily through childhood, peaks in early adulthood, and then declines slowly over decades. However, not everyone reaches the same peak or declines at the same rate. Some individuals follow high, stable lung-function trajectories throughout life. Others fail to achieve optimal lung development due to early-life exposures such as prematurity, recurrent childhood infections, secondhand smoke, or air pollution. These individuals enter adult life with a lower respiratory reserve and remain on a disadvantaged trajectory even if symptoms improve.This early-life divergence has profound consequences. Research shows that suboptimal lung trajectories predict earlier cardiovascular disease, more systemic inflammation, depression, metabolic dysfunction, and premature frailty. In other words, the lungs quietly set the stage for the body’s capacity to withstand the challenges of aging long before the tipping point of 75 arrives.
Why Aging Can Feel Like “Everything Happens at Once”
The heart, lungs, immune system, and musculoskeletal system do not operate independently. They influence one another through shared physiological pathways, particularly inflammation and oxygen delivery. When lung function falters, oxygenation decreases and inflammatory signals rise. These changes place extra strain on the heart, which in turn accelerates fatigue and activity limitations. Reduced activity worsens muscle loss, which amplifies frailty and increases vulnerability. Over time, the accumulation of these deficits creates a feedback loop in which each new problem makes the next more likely. This “damage promotes damage” phenomenon helps explain why health often appears to deteriorate suddenly in older adults.
The Gender Paradox in Frailty and Longevity
One of the more puzzling observations in aging research is that men tend to appear less frail in early life yet die sooner than women. The dynamical model of frailty offers insight into this paradox. Men begin adulthood with greater robustness and resilience, but these advantages decline more rapidly. Women accumulate more deficits but maintain survival advantages, in part due to slower rates of physiological decline. As a result, men typically reach the frailty tipping point earlier and with fewer reserves to buffer subsequent damage.
Changing the Trajectory: A New Vision for Healthy Aging
Perhaps the most important insight from these studies is that the trajectory leading toward the age 75 tipping point is not fixed. Health trajectories can be bent, gradually, but meaningfully, through interventions that strengthen resilience and reduce inflammation. Early life airway protection, clean indoor air, avoidance of tobacco smoke, and treatment of allergic or inflammatory conditions can set the stage for healthier lung trajectories. In midlife, regular exercise, strength training, optimal nutrition, and control of cardiovascular risk factors preserve robustness and slow the rate of deficit accumulation. After 75, protecting the body from stressors becomes essential, whether through fall prevention, vaccination, good air quality, or rapid treatment of respiratory infections.
A New Framework for Understanding Aging
The convergence of frailty modeling, cardiac aging, and lung-trajectory research provides a clearer and more holistic framework for understanding aging. Health is not simply something we possess, but something we travel along, a trajectory shaped by childhood exposures, lifelong inflammation, environmental factors, and organ-specific aging. The tipping point at 75 represents a predictable phase transition in physiology, but the timing and severity of that transition are modifiable.
This scientific view reframes aging not as an unavoidable decline but as a dynamic process in which early intervention, continual monitoring, and sustained attention to airway and cardiovascular health can delay frailty, extend independence, and improve quality of life well into advanced age.
Reference
1. Shinmura K. Cardiac Senescence, Heart Failure, and Frailty: A Triangle in Elderly People. Keio J Med. 2016;65(2):25-32. doi:10.2302/kjm.2015-0015-IR
2. Pridham G, Rockwood K, Rutenberg AD. Dynamical modelling of the frailty index indicates that health reaches a tipping point near age 75. arXiv. Published December 2, 2024. arXiv:2412.07795. doi:10.48550/arXiv.2412.07795
3. Melén E, Faner R, Allinson JP, et al. Lung-function trajectories: relevance and implementation in clinical practice. Lancet. 2024;403(10435):1494-1503. doi:10.1016/S0140-6736(24)00016-3